|
Mendeleev was unable to accurately assign hydrogen's location in the periodic table.These elements are now known as Sc, Ga, and Ge. He also anticipated their qualities as well as their atomic masses. He called the hitherto unknown elements Eka-Boron, Eka-Aluminium, and Eka-silicon. There were many empty slots in Mendeleev's Periodic table for additional element discovery.Mendeleev's Periodic table corrects various atomic masses of elements.Mendeleev's Periodic Table has the following advantages: Vertical columns are referred to as 'groups,' while horizontal rows are referred to as 'periods,' in Mendeleev's Periodic Table. 
He discovered a link between the elements' atomic masses and their physical and chemical qualities, as well as a periodic recurring trend with comparable physical and chemical attributes. He began his investigation with just 63 known elements. Mendeleev's Periodic Table: In 1869, Russian scientist Dmitri Ivanovich Mendeleev developed a Periodic rule. As a result, the remaining components and their attributes did not adhere to the law of octaves. When Newland issued this legislation, there were only 56 elements in nature, but since then, many elements have been found. The disadvantages of the octaves law are that it is only relevant up to Ca, and after Ca, every eighth element does not have attributes identical to the first. As a result, it is known as Newland's Law of Octaves. He linked these parallels to musical octaves. He found a pattern in every eighth element that had qualities identical to the first one. The Law of Octaves by Newland: In 1866, John Newlands, an English chemist, organized the 56 known elements in order of increasing atomic mass, beginning with hydrogen and ending with thorium. The disadvantages of Dobereiner's Triads were that these were inapplicable to elements with extremely high and low atomic masses, and this categorization was not applicable to all of the elements. Na, the middle element in this triangle, has an atomic mass of 22.99. As a result, these groupings are known as 'triads.' For example, Li has an atomic mass of 6.94, while K has an atomic mass of 39.10. He demonstrated that three elements might be arranged in a triad so that the atomic mass of the middle element is about equal to the average of the atomic masses of the other two elements. 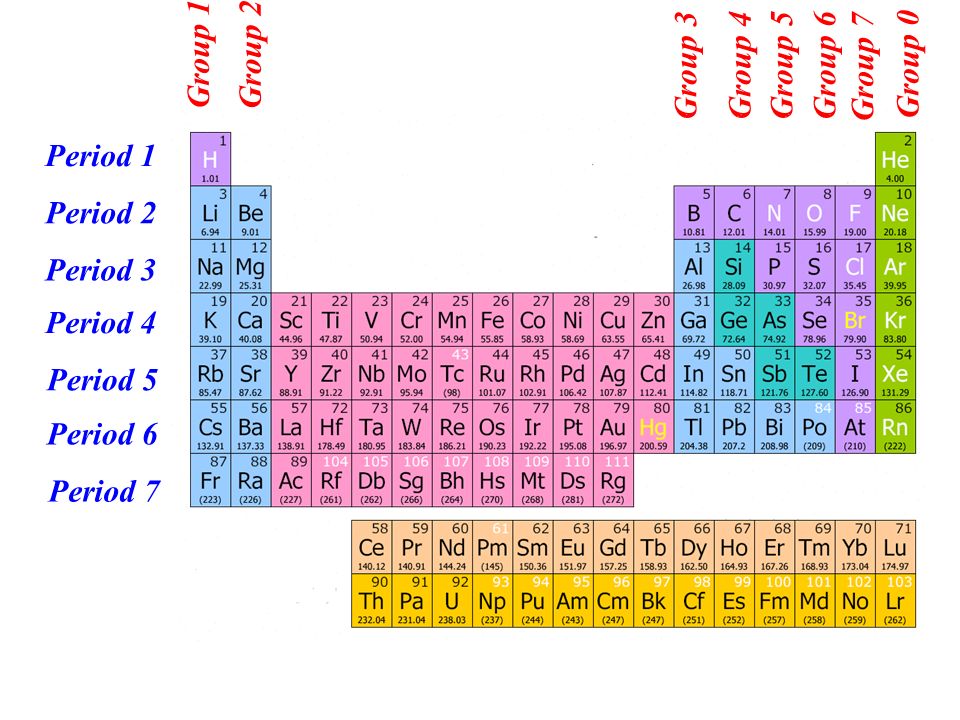
The Origins of Periodic Classificationĭobereiner's Triads: In 1817, Johann Wolfgang Dobereiner, a German scientist, attempted to organize substances having similar characteristics into groups. Scientists are looking for a systematic technique to rearrange and organize knowledge by categorizing it in order to solve this dilemma. As a result, studying the chemistry of all the different elements is extremely challenging. In the beginning, just 31 elements were known, but currently, 118 elements are known.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
